| Table of Contents |  |
|
Original Article
| ||||||
| Face and emotional expression processing and event-related potentials in a case study of impaired face perception | ||||||
| Lucy J. Troup1, Stephanie Bastidas2, Jason S. Nomi3, Maia T. Nguyen2, Tien Tong4 | ||||||
|
1PhD, Colorado State University, Department of Psychology, Colorado State University, Fort Collins, CO, United States.
2MS, Colorado State University, Department of Psychology, Colorado State University, Fort Collins, CO, United States. 3PhD, University of Miami, Department of Psychology, University of Miami, Miami, Florida, United States. 4BSc, Colorado State University, Department of Psychology, Colorado State University, Fort Collins, CO, United States. | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article]
[Similar article in Pumed] [Similar article in Google Scholar] |
| How to cite this article |
| Troup LJ, Bastidas S, Nomi JS, Nguyen MT, Tong T. Face and emotional expression processing and event-related potentials in a case study of impaired face perception. Edorium J Psychol 2015;1:9–17. |
|
Abstract
|
|
Aims:
To evaluate face and emotional expression recognition in a single participant event related potential (ERP) case study.
Methods: We compared an individual with impaired face perception (participant G.O.) to 29 normal controls in behavioral tests of object, face and expression recognition and also recorded Event-Related Potentials (ERPs) in response to houses, faces and faces with emotional expressions. Results: Participant G.O. performed normally on behavioral tests of object and emotional expression recognition but was significantly impaired in tests of face recognition. Unlike controls, G.O. did not show a difference in mean amplitude of P1 to houses compared to upright faces. Additionally, G.O. presented with a marked amplitude decrease in the temporal occipital N170 in response to faces compared to controls and a decrease in N170 and P300 amplitude in response to emotional expressions compared to controls. G.O. clearly showed a unique pattern of face and emotion recognition compared to control participants. Conclusion: The behavioral deficits were not directly reflected in the ERP responses found for G.O. and controls. However, G.O. presented a distinctive pattern of scalp electrical activity for faces, both standard and with emotion. Thus highlighting the importance of using multiple measures in the examination of face perception deficits in individuals | |
|
Keywords:
Event related potentials, Face recognition impairment, Emotion
| |
|
Introduction
| ||||||
|
Human face recognition is a vastly complex process that on initial inspection appears to be seamless. Within the literature addressing human face perception there is consensus that there are brain structures both independent and overlapping that are necessary for face processing. Imaging studies such as positron emission tomography (PET) scan and functional magnetic resonance imaging (fMRI) scan, identify the fusiform gyrus, in the dorsal stream as a structure central to face perception [1] [2] [3] [4] [5] [6]. Other imaging techniques such as event-related potential (ERP) have focused attention on temporal markers that represent face perception [7]. Explanations of face perception, including those which account for recognition in terms of identity and expression, capture both the serial, and parallel nature of these structural accounts. The Bruce and Young (1986) model of face recognition and Haxby, Hoffman, and Gobbini (2000) distributed model of face recognition hypothesize independent mechanisms for the processing of identity and expression; Bruce and Young propose independent but parallel processing of identity and expression while Haxby et al. propose separate but interacting mechanisms for identity and expression recognition a name="ft5" href="#ref5">[5] [8]. The two models are supported by evidence finding identity recognition and expression recognition rely on independent and overlapping cortical networks [9][10][11][12][13]. Individuals with face perception deficits such as prosopagnosia, the inability to recognize faces, demonstrate impairments in behavioral face recognition tasks while performing normally on behavioral tasks of emotional expression recognition [13] [14] [15] [16] [17] [18]. Additionally, individuals with these deficits demonstrate a marked decrease in the amplitude of the ERP most associated with face perception, the temporal-occipital N170. In normal individuals the N170 amplitude is much larger in response to faces compared to other objects such as cars or houses, whereas there is typically no difference between faces and objects for individuals with prosopagnosia [7] [19]. Therefore, the poor performance by individuals with prosopagnosia in behavioral tests of face recognition could be attributed to similar amplitude for the N170 in response to faces and houses. However, it is unclear in literature how emotional expressions influence ERPs in individuals with developmental prosopagnosia compared to normal controls. Although individuals with prosopagnosia perform normally on behavioral tests of expression recognition, it is still not clear if their ERPs for emotional expression recognition match those of normal controls. Studies have found that emotional expression modulates the amplitude of the N170, while others have found no expression influence on the N170 amplitude [20] [21]. Additionally, emotional images have been shown to influence the Late Positive Potential (LPP) ERP, or the P300, in normal individuals but there is a deficit in the literature regarding this effect in individuals with prosopagnosia [22]. Therefore, it is unclear how emotional expressions will influence the N170 and P300 in individuals with developmental prosopagnosia and normal controls. Aims | ||||||
|
Materials and Methods
| ||||||
|
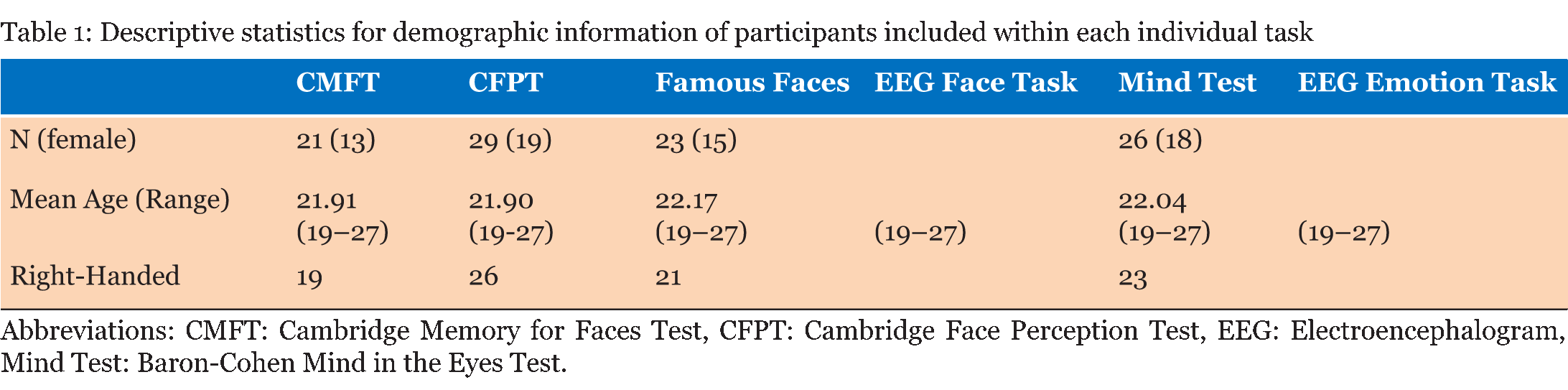
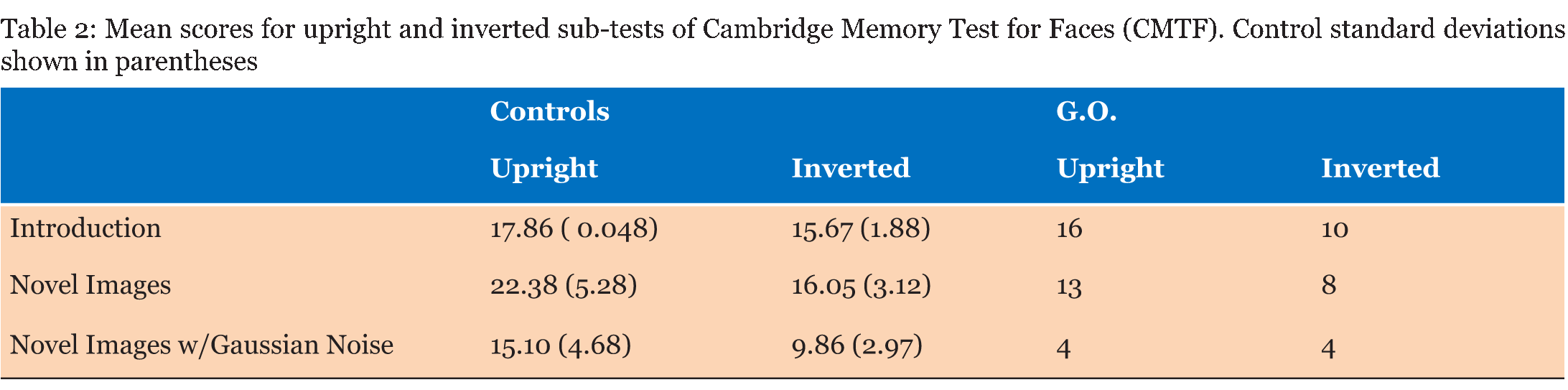
Case Description Neurologically Intact Controls: Twenty-nine undergraduate students (19 females; ages 19–27) with no history of neurological illness and no significant symptoms of depression or anxiety were included in the control group. Although our controls were not aged matched, literature is conflicted as to the effects that age has on ERP's specifically that difference are mostly seen in latency of the component. Evidence from literature suggests differences are very much task specific, and do not have a significant effect on sensory processing [23] [24]. The N170 component for example is considered relatively stable over age in adulthood [25]. The P300 does exhibit some age and sex related differences that are indicative of normal aging and cognitive decline, although the decrease in amplitude and increase in latency are gradual [26]. The number of participants who fully completed individual tasks varied, ranging from 21 to 29; descriptive for each sub-group are given in (Table 1). All students received credit in a Psychology course for their participation and provided written consent. Face Perception Tasks Cambridge Memory for Faces Test: Developed by Duchaine and Nakayama (2006), this test examined recognition memory for faces in a forced-choice paradigm [27]. Grayscale photographs of 52 males in their 20s and 30s from three different views (frontal, left 1/3 profile, and right 1/3 profile) with cropped hair were used. Pictures of six individuals represented target items and the remaining 46 individuals represented distractors, grouped as one target and two distractors per test item. Practice stimuli consisted of three views (frontal, left 1/3 profile, and right 1/3 profile) of Bart Simpson. Participants completed an upright version and an inverted version with three stages of testing each: an introductory stage in which each target item was tested separately, and two novel image stages, one with Gaussian noise added. During practice and introduction, three views were presented of each test for 3000 ms each, followed by three test items in which participants pressed a key to identify the target for a total of three practice and 18 introduction trials. In the novel images condition, all six faces were presented simultaneously for 20s in a frontal view for study, followed by 30 test items presented at different angles (6 targets x 5 tests). The novel images with noise condition had Gaussian noise added to test images and there were 24 test items (6 targets x 4 tests). Reaction time and accuracy were recorded for each stage. Cambridge Face Perception Test: The CFPT for impairments in perception of upright and inverted faces; participants are presented with morphed faces that they must order based on similarity to a target face [28]. Grayscale images of six male subjects at a ¾ profile view represented target items; comparison items consisted of frontal views morphed towards images of other subjects by 28%, 40%, 52%, 64%, 76%, and 88%. Participants completed 2 practice trials (1 inverted) and eight upright and eight inverted trials, presented in randomized order. Scores represented the average number and percent errors for the upright and inverted conditions, calculated as deviation from the correct position for each face. Famous Faces Test: Thirty photographs of celebrities and politicians with cropped hair were presented on a computer screen at a viewing distance of 30 cm [9]. Each face was presented for 3000 ms after which participants were allowed unlimited time to provide the name of the celebrity or, if unknown, to write down any information unique to the depicted individual. Correct responses entailed providing the name of or information unique to the individual (e.g., "Captain Jean-Luc Picard" for Sir Patrick Stewart). EEG Face Task: Stimuli consisted of grayscale images of 16 faces (8 females) with cropped hair measuring 239x276 pixels presented upright and inverted, and 16 houses measuring 368x276 pixels shown on a black background. As an attention check, participants were instructed to press a key every time a white box (276x276 pixels) appeared on the screen. Each block contained one presentation of each stimuli for a total of 48 trials presented in random order. Each trial consisted of delay of 1300 ms during which a black screen was presented, followed by a 100 ms presentation of an upright or inverted face or a house. Controls completed 8 blocks while G.O. completed 16 blocks of the task. Emotion Perception Tasks EEG Emotion Task: Stimuli were grayscale images of 20 participants (10 females) depicting three facial expressions (Happy, Sad, Neutral; NimStim face database), presented with hair obscured by a black oval mask, and resized to 210x270 pixels [30]. Based on conditions from Rellecke, Sommer, and Schacht (gender decision, emotional decision), the explicit emotion processing condition consisted on explicit decisions about the emotional expression of the portrayed person, while the implicit emotion processing condition consisted on decisions about the sex of the person [31]. Stimuli were separated into two sets of 30 counterbalanced by task assignment: half of the participants viewed set 1 during implicit processing and set 2 during explicit processing, and vice versa. Each trial started with an instruction prompt to attend to either the Sex or the Emotion depicted by the individual, corresponding to implicit and explicit processing of the facial expressions. The instructional prompt (2000 ms) was followed by 1500 ms allowed to internalize the instructions, then a fixation cross (1000 ms) preceded the expression image, shown for 2000 ms. Finally, the participant was allowed up to 2000 ms to press a key with the appropriate response (implicit: male or female; explicit: neutral, happy, or angry). General Procedure EEG Acquisition: Electroencephalography (EEG) was recorded from 21 Ag/AgCl electrodes (midline: Fz, Cz, Pz; left: Fp1, F3, F7, C3, T7, P3, P7, PO7, O1; and corresponding right electrodes) mounted on a SynAmps2 64-channel QuikCap (CompumedicsNeuroScan, Charlotte, NC, USA) according to the 10-20 system. The vertex was used as online reference. Horizontal electrooculogram was monitored with electrodes placed on the outer canthi of the left and right eyes. Impedance was kept below 11 O. Signals were recorded at a sampling rate of 500 Hz and amplified with a band pass of .10-50 Hz in epochs from -200 to 400 ms for the face processing task and -200 to 1000 ms for the emotion processing task. Data Analysis: Average reaction times and percent correct responses during each task were obtained for all participants. EEG data was re-referenced offline to the common average and baseline corrected to pre-stimulus period. Artifact rejection was applied to trials with amplitudes exceeding ±50 µV at HEO channels and ±100 µ at remaining electrodes. Grand averages for the face processing task were examined in ANOVAs for controls by Type (Face-Upright, Face-Inverted, House) x Electrode (parietal, parieto-occipital, occipital)xHemisphere (left, right), based on mean amplitudes and peak latencies were for P1 (80–140 ms), N170 (140–200 ms), and P3a (200–400) components. The emotion processing task was similarly analyzed by Task (Implicit, Explicit), Emotion (Angry, Neutral, Happy), Electrode (temporal, parieto-occipital, occipital), and Hemisphere (left, right) with the addition of P3b amplitude and latency (400–600 ms). Latencies were the time in milliseconds corresponding to the peak amplitude for each component. Significant differences were further investigated using paired sample t-tests. Differences between G.O. and controls were examined using Crawford and Garthwaite modified t-test for single case studies [34]. Alpha levels were set at α=0.05 with a Bonferroni correction for post-hoc tests where appropriate. Participants with >90% "no responses" or >75% rejected EEG trials were excluded accordingly, resulting in variations in Control group makeup for each task (Table 1). | ||||||
| ||||||
|
Results | ||||||
|
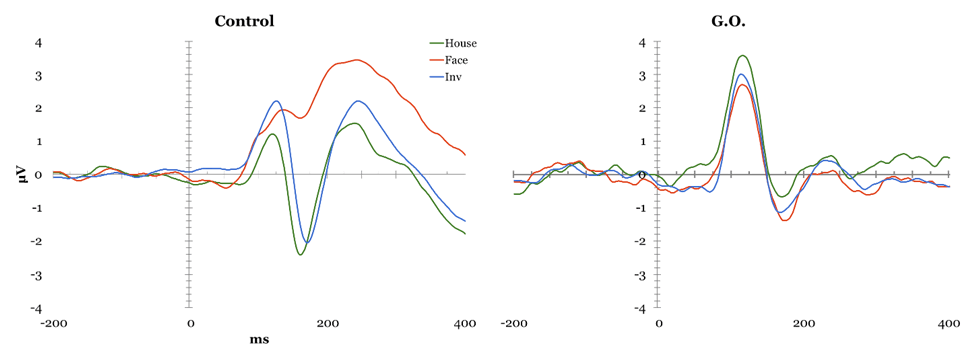
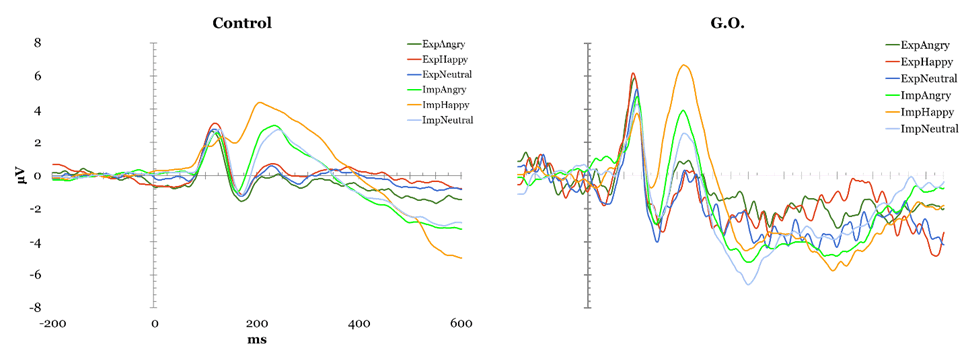
Face Perception Tasks Cambridge Face Perception Test: Scores on the CFPT reflect deviations from the correct position for each face added across all items for each orientation, with higher scores representing a greater number and percent of errors. Overall, controls showed an inversion effect, with a higher number of errors in the inverted condition (M = 39.52, SD = 9.527) than in the upright condition (M = 66.48, SD = 12.429), t(28) = -8.954, p < 0.01. G.O.'s error scores did not differ from controls' during upright trials (54 vs. M = 39.52, SD = 9.527), t(28) = 1.495, p > 0.05, or inverted trials (56 vs. M=66.48, SD = 12.429), t(28) = -.829, p > 0.05. Famous Faces Test: G.O.'s score of 4 correct out of 30 presented faces was lower than control participants' mean score 19.78 out of 30, t(22) = -2.32, p < 0.05. EEG Task: All participants obtained over 90% correct detections on the attention check and averaged 37.49% rejected ERP trials, with one participant being excluded for excessive rejected trials (100%). The remaining control participants were compared to G.O. (Figure 1). P1: A main effect of electrode was found, F(2, 46) = 6.644, p < 0.05, such that P1 mean amplitude was greater at parieto-occipital than occipital, t(143) = 2.996, p < 0.01, and temporal sites, t(143) = -4.36, p < 0.01. A significant type by electrode interaction, F(4, 92) = 8.012, p < 0.05, suggested controls showed smaller mean amplitude for houses compared to upright, t(47) = 3.244, p < 0.01, and to inverted faces, t(47) = -4.13, p < 0.01 at parieto-occipital electrodes but not at other sites. This pattern was not observed in G.O. No P1 latency effects were identified. N170 mean amplitude: Controls presented an effect of Type, F(2, 46) = 24.057, p < 0.05, with smaller N170 amplitude for houses than upright, t(143) = -9.787, p < 0.01, and inverted faces, t(143) = 12.468, p < 0.01; and for upright than inverted faces, t(143) = 5.436, p < 0.01. A main effect of electrode, F(2, 46) = 14.642, p < 0.05, showed greater mean N170 amplitude at temporal than parieto-occipital, t(143) = -6.888, p < 0.01, and occipital sites, t(143) = -8.175, p < 0.01, and at parieto-occipital than occipital sites, t(143) = -3.357, p < 0.01. A significant type by electrode interaction, F(4, 92) = 3.224, p < 0.05, suggested these differences were largest at parietal sites, followed by parietooccipital, and smallest at occipital sites. G.O. showed no change in performance on houses and inverted faces, but a decrease in performance for upright faces most noticeable at parietooccipital sites. N170 Latency: A main effect of Type was found, F(2, 46) = 22.942, p < 0.05, showing greater N170 latency for inverted than upright faces, t(143) = -8.253, p < 0.01, and houses, t(143) = -10.383, p < 0.01, and for upright faces than houses, t(143) = 5.688, p < 0.01. A main effect of Electrode, F(2, 46) = 8.253, p < 0.05, showed shorter latency at occipital than temporal, t(143) = 4.135, p < 0.01, and parieto-occipital sites, t(143)=4.565, p < 0.01, while G.O. showed largest latency at parieto-occipital sites. A significant type by electrode interaction was found, F(2.194, 50.456)=9.457, p < 0.01, with greater latency for inverted faces than upright faces and houses, and for upright faces than houses at all electrode sites except for temporal, t(47)= 0.0588, p > 0.05. while G.O. showed a similar pattern over occipital sites, with no differences between inverted and upright faces. Overall, G.O. presented later N170 for upright, t(24)=1.924, p < 0.05, and inverted faces, t(24) = 1.918, p < 0.05, but not houses, t(24) = 1.595, p > 0.05, compared to controls. P3a Maximum Amplitude: A significant effect of Type, F(2, 46) = 18.503, p < 0.05, suggested reduced P3a for houses compared to upright and inverted faces, both in Controls and G.O. An effect of Electrode, F(2, 46) = 8.144, p < 0.05, with smallest P3a over temporal electrodes, was modulated by an Electrode by Hemisphere interaction, F(2, 46) = 3.194, p < 0.05, in which P3a was reduced over left temporal compared to left parieto-occipital and left occipital electrodes, with no differences over right sites or between hemispheres. G.O. presented a similar pattern with the addition of reduced P3a also over parieto-occipital left sites. P3a Latency: An effect of electrode, F(2, 46) = 5.961, p < 0.05, suggested faster P3a over occipital sites in Controls, which G.O. also presented. Further, a significant effect of Type, F(2, 46) = 3.306, p < 0.05, showed faster P3a for upright faces compared to houses and to inverted faces, while G.O. presented a slower P3a only for inverted faces. Emotion Processing Tasks EEG Emotion Processing Task: Participants averaged 49.32% rejected trials; three participants being excluded for excessive rejected trials (99.56–100%). Data from remaining controls was compared to G.O.'s (Figure 2). P1 amplitude: A main effect of electrode, F(2, 42) = 7.687, p < 0.05, showed smaller P1 mean amplitude at temporal than parieto-occipital and occipital sites in controls. G.O. presented a similar distribution with no differences in amplitude from controls at any electrode sites (temporal: t(24) = -0.202, p > 0.05; parieto-occipital: t(24) = 0.408, p > 0.05; occipital: t(24) = 0.097, p > 0.05). No effects of P1 latency were found. N170 amplitude: A main effect of electrode, F(2, 42) = 7.545, p < 0.05, found greater mean amplitude at temporal than parieto-occipital, t(263) = -5.425, p < 0.01, and occipital sites, t(263) = -6.311, p < 0.01. G.O. presented a different pattern in which N170 amplitude was largest at parieto-occipital rather than temporal sites. A Task by Emotion by Hemisphere effect, t(2, 42) = 4.31, p < 0.05, highlighted greater right than left N170 for Happy stimuli overall, and for Neutral stimuli only during implicit processing. G.O. presented reversed patterns based on task: larger N170 over right electrodes for explicit processing of neutral and implicit processing of happy; this was also present for angry faces overall. N170 latency: A significant effect of emotion, F(2, 42) = 3.499, p < 0.05, suggested slower N170 for neutral than happy and angry faces in controls, whereas G.O. showed greatest latency for angry faces, t(263) = 3.919, p < 0.01, followed by neutral, t(263) = 1.392, p < 0.01, then happy faces, t(263) = -2.685, p < 0.01. G.O.'s N170 was slower overall compared to controls', t(24) = 1.709, p > 0.05. P3a Amplitude: Effects for Electrode were found, F(2, 42) = 3.485, p < 0.05, with smallest maximum amplitude over temporal sites, followed by occipital, and maximum amplitude over parieto-occipital sites. G.O. presented no differences from controls. P3a Latency: A Task by Emotion interaction showed shorter latency for Happy than Angry faces during explicit processing that was reversed during explicit processing, F(2, 42)=3.492, p < 0.05.; while G.O. showed slower P3a for Happy faces independent of task, reflected as overall slower P3a compared to controls, t(24)=-2.441, p < 0.05. P3b Amplitude: In controls, a Task by Emotion by Electrode interaction trended towards significance F(4, 84) = 2.328, p=0.061, suggesting greater amplitude for explicit processing of Angry faces over temporal electrodes and Happy faces over parieto-occipital electrodes. G.O. did not present differences by emotion and electrode site. No latency effects were identified. | ||||||
| ||||||
| ||||||
| ||||||
|
| ||||||
| ||||||
|
Discussion
| ||||||
|
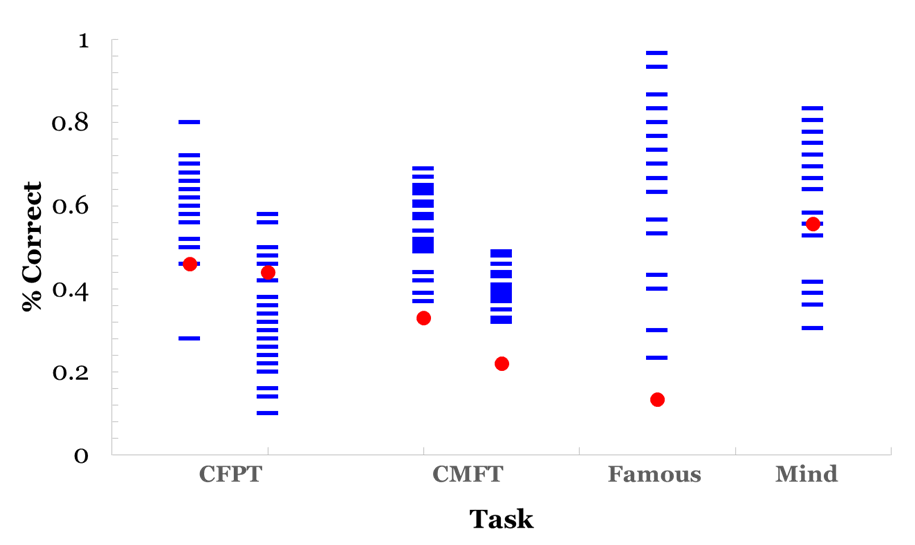
Participant G.O. showed clear deficits in face recognition in behavioral and ERP responses, accompanied by differential patterns in emotion processing most marked in ERP than behavioral measures. G.O. scored significantly lower than controls on measures of recognition for studied new faces (Cambridge Memory for Faces Task) and for unstudied famous faces. His behavioral deficits in processing appear to be limited to face recognition ( (Figure 3), left), as he presented no deficits in face perception (Cambridge Face Perception Test), or in forced-choice measures of emotion identification (Mind in the Eyes) and discrimination ( (Figure 3), right). This pattern of response is not necessarily indicative of prosopagnosia but it is consistent with prosopamnesia: he is able to perceive faces but has difficulties encoding and retrieving specific faces [35]. Prosopamnesia is a condition in which memory for faces is selectively impaired. Although G.O was able to perceive faces normally, there was a clear deficit in his memory for faces and his ability to maintain recognition over time. This is consistent with face recognition impairment and similar to performance on face memory tasks for individuals with a diagnosis of prosopagnosia and prosopamnesia. It is noted in the literature that it is often difficult to distinguish clinically between the two conditions [36]. His ability to recognize and process emotional expression remained intact despite his face recognition deficits. ERP responses The results suggest an interesting pattern in which G.O. does not present behavioral deficits engaging in emotional expression processing, but he possibly relies on different mechanisms from controls in processing emotional expression. This differs from the intact emotional expression recognition reported in developmental prosopagnosia [9] [11]. G.O.'s performance supports aspects of both of the dominant models of face and emotional expression recognition [5] [8]. In relation to the Bruce and Young explanation, emotion expression recognition and face recognition occur in parallel in separate systems in a linear fashion. Haxby et al.'s more recent model suggests that although there is independence of emotional expression and identity recognition the two processes are both independent and integrated. G.O. clearly has deficits in one system (face recognition) that is still intact in another (emotional expression recognition) consistent with the Bruce and young account, but the subtleties of his deficits in emotional expression recognition suggest that the independence of these deficits are not as clear and favor the Haxby et al. account [5] [8]. Differences in scalp distribution during familiarity and recollection has suggested the use of "abnormal routes" to face recognition in DPs [37]; the extent of the deficits may contribute differently to the routes used for expression processing later in life. While it remains to be established how common dissociations between behavioral and neurophysiological deficits are face processing impairments, this topic might provide further support to the relationship between impaired identity and expression recognition [10]. Neither the Haxby et al. nor the Bruce and Young models provides a complete explanation for the differences between behavioral and neuronal performance that we see in participant G.O. [5] [8].The Bruce and Young model could explain the behavioral results of impaired identity recognition with intact expression recognition, but the Haxby model would allow for the differences observed in both N170 and P300 ERPs to expression processing. Previous research has demonstrated that emotional expression influences the N170, therefore, G.O.'s marked decrease in N170 could result in the noted decrease in the P300 [20]. That is, G.O.'s deficit in face recognition as reflected in his decreased N170 could possibly be driving the ERP differences during emotion recognition reflected in the P300. This still does not explain his normal behavioral responses to emotional expression. Our results support a model of face and emotional expression recognition that suggests these processes are somewhat independent but highly related [5]. Face and expression processing is modular yet there is interaction between the processing of both a given face and its expression. | ||||||
|
Conclusion
| ||||||
|
It is important to note that there is variance in deficits among individuals with face perception impairments, whether developmental or acquired. Further, while this ERP data might reflect the processing mechanisms of face and emotional expression recognition, it does not necessarily mean that those processing differences are related to similarities in the underlying functional architecture. As discussed, the same brain areas might not be responsible for the deficits we are observing in different individuals. We are currently following up this study with two more prosopagnosics to compare their results with those of G.O. as well as including controls that were more closely aged matched. Although we were able to use controls who were in a closer age range to G.O. there was still a deficit in years that we will address in future work. | ||||||
|
Acknowledgements
| ||||||
|
Sponsored by the National Science Foundation (NSF) Research Experiences for Undergraduates (REU) Program, NSF Grant SMA-1005199 to Edward L. DeLosh. | ||||||
|
References
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions:
Lucy J. Troup – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Stephanie Bastidas – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Jason S. Nomi – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Maia T. Ngyuen – Substantial contributions to conception and design, Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Tien Tong – Substantial contributions to conception and design, Acquisition of data, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of submission
The corresponding author is the guarantor of submission. |
|
Source of support
None |
|
Conflict of interest
Authors declare no conflict of interest. |
|
Copyright
© 2015 Lucy J. Troup et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|